EU Medicines Agency on X: "#EMA supports #RareDiseaseDay every day! Our Committee for Orphan Medicinal Products is the only committee fully dedicated to medicines for rare diseases. #ShowYourRare https://t.co/uNumz8tDDX https://t.co/heMlVfyYnm" / X
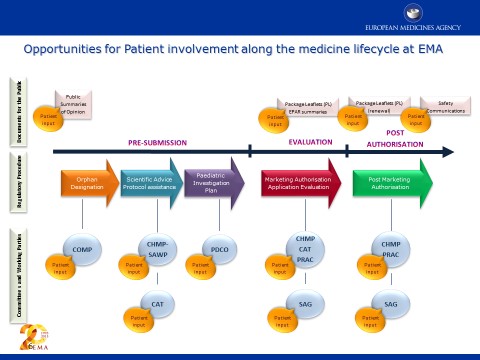
Public summary of opinion on orphan designation Cisplatin for the treatment of malignant mesothelioma
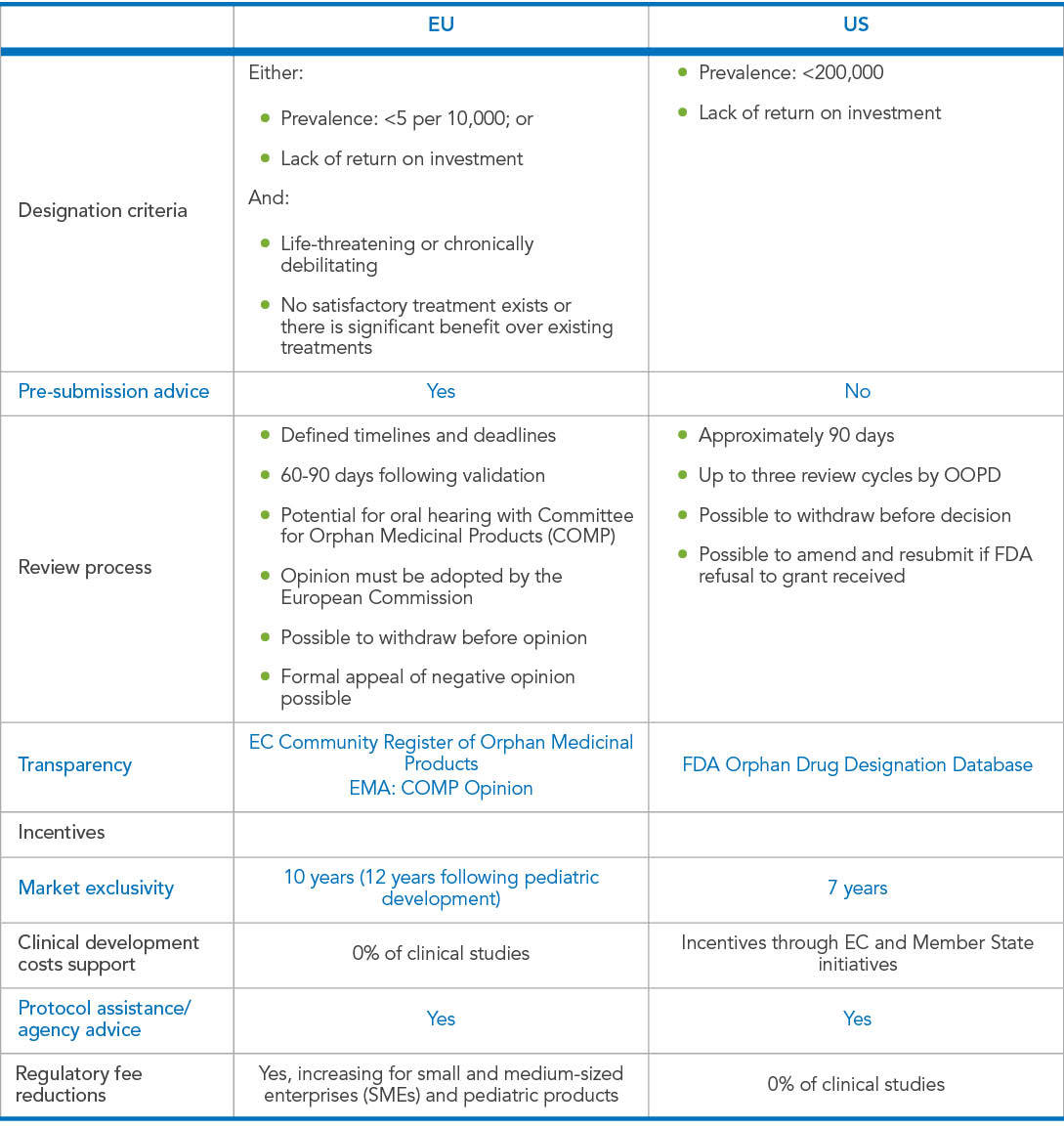
Perspective Paper: Four Considerations to Shape Your Orphan Drug Designation Application Strategy - Evidera
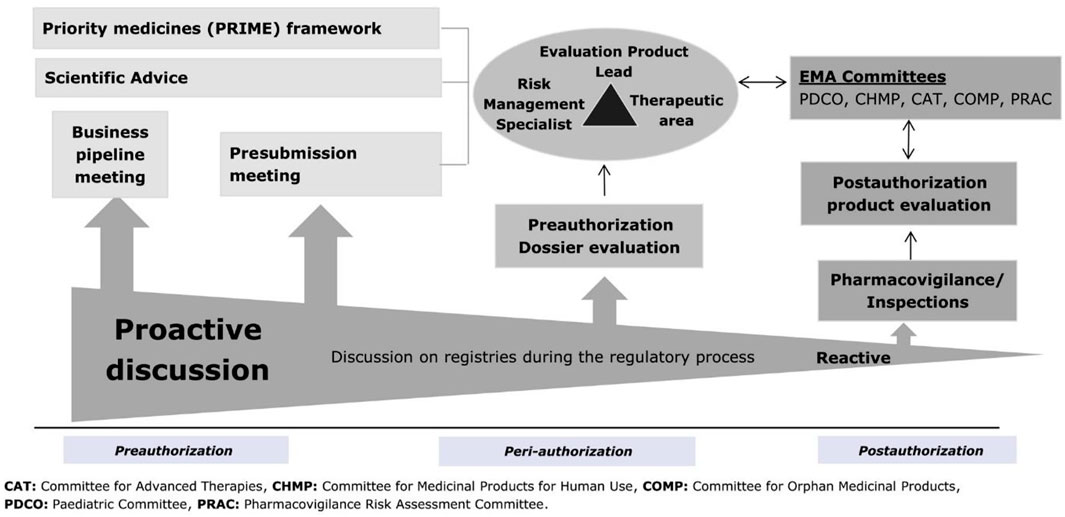
Frontiers | Contribution of patient registries to regulatory decision making on rare diseases medicinal products in Europe

Use of biomarkers in the context of orphan medicines designation in the European Union – topic of research paper in Clinical medicine. Download scholarly article PDF and read for free on CyberLeninka