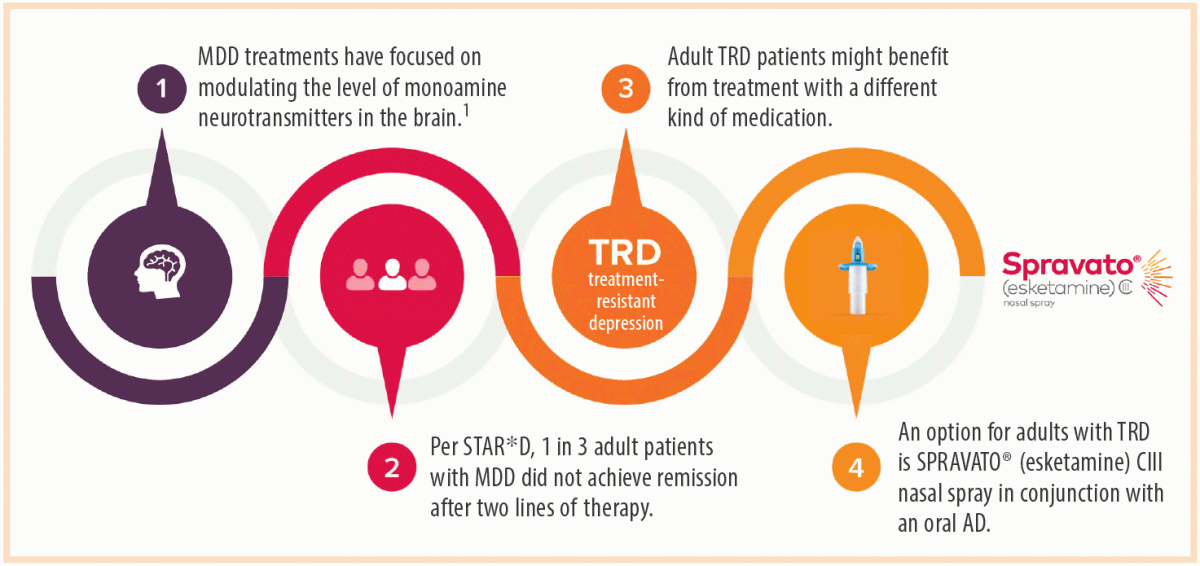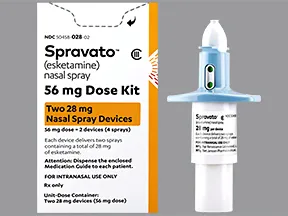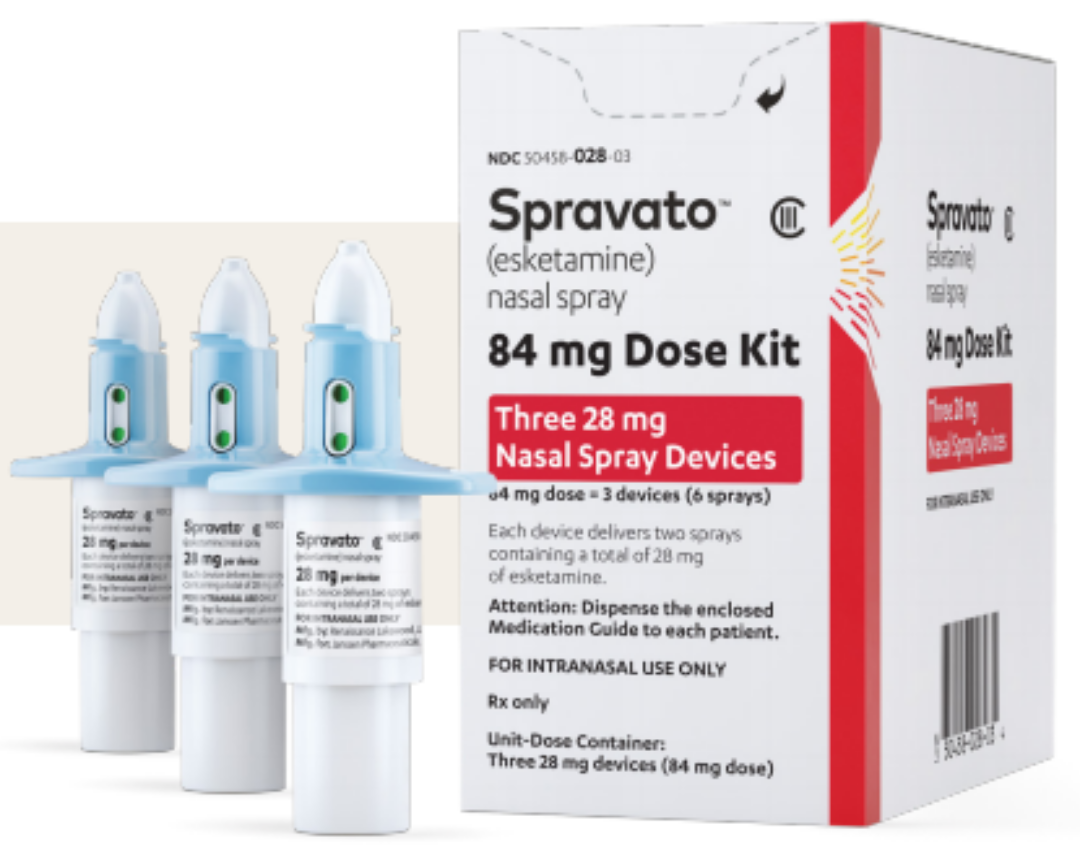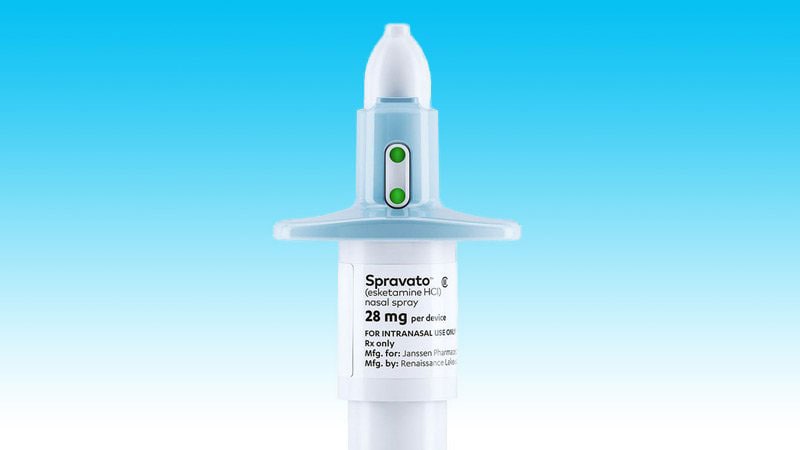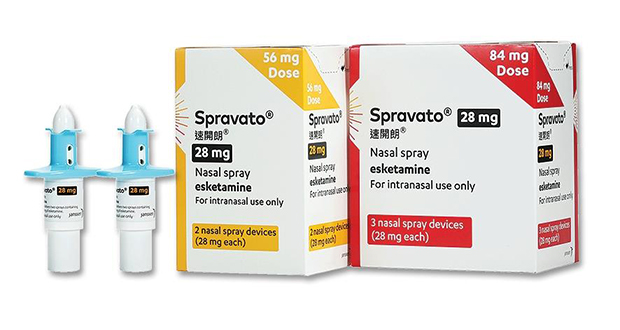
SPRAVATO®▽ approved for reimbursement in Ireland for adults with treatment-resistant major depressive disorder

Janssen Announces U.S. FDA Approval of SPRAVATO® (esketamine) CIII Nasal Spray to Treat Depressive Symptoms in Adults with Major Depressive Disorder with Acute Suicidal Ideation or Behavior